Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Hirade, Tetsuya; Michishio, Koji*; Kobayashi, Yoshinori*; Oshima, Nagayasu*
Chemical Physics Letters, 795, p.139507_1 - 139507_4, 2022/05
Times Cited Count:0 Percentile:0.01(Chemistry, Physical)We obtained the temperature dependence up to 150 C of the triplet positronium (
C of the triplet positronium (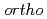 -Ps) lifetime in N,N,N-Trimethyl-N-propylammonium bis(trifluoromethanesulfonyl)imide (TMPA-TFSI) by the vertical slow positron beamline installed at AIST. Positrons penetrate into the liquid surface of TMPA-TFSI with the positron energies of 2 keV and 12 keV to investigate at the near-surface and the balk. The surface structure was visible at 150
-Ps) lifetime in N,N,N-Trimethyl-N-propylammonium bis(trifluoromethanesulfonyl)imide (TMPA-TFSI) by the vertical slow positron beamline installed at AIST. Positrons penetrate into the liquid surface of TMPA-TFSI with the positron energies of 2 keV and 12 keV to investigate at the near-surface and the balk. The surface structure was visible at 150 C, 120
C, 120 C above the melting temperature. The
C above the melting temperature. The 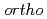 -Ps lifetime became shorter at higher temperatures for both positron energies. Similar temperature dependence had appeared just in water as the result of the reaction of
-Ps lifetime became shorter at higher temperatures for both positron energies. Similar temperature dependence had appeared just in water as the result of the reaction of 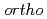 -Ps and radiolysis products such as the OH radicals. The temperature dependence observed for TMPA-TFSI suggested that the chemical reaction of
-Ps and radiolysis products such as the OH radicals. The temperature dependence observed for TMPA-TFSI suggested that the chemical reaction of 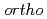 -Ps occurred.
-Ps occurred.
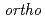 -positronium and radical in liquids
-positronium and radical in liquidsHirade, Tetsuya
Proceedings of 8th Asia Pacific Symposium on Radiation Chemistry (APSRC 2020) (Internet), 2 Pages, 2020/04
The positrons injected in insulating materials make ionization at the end part of the track, forming excess electrons and radicals. When one of the excess electrons and the incident positron form positronium, the electron in the positronium and the unpaired electron in the radical have a spin correlation. By using this spin correlation, a quantum beat phenomenon that depends on the hyperfine coupling constant of the radical appears in the spin conversion reaction with the positronium. The spin-correlated radical and other radicals can be distinguished by a reaction using the positronium as a probe. It is possible to study the behavior of OH radicals in water and the state of cation radicals in room temperature ionic liquids.
Hirade, Tetsuya; Michishio, Koji*; Kobayashi, Yoshinori*; Oshima, Nagayasu*
Acta Physica Polonica A, 137(2), p.109 - 112, 2020/02
Times Cited Count:0 Percentile:0(Physics, Multidisciplinary)Recently, it was reported that the positronium (Ps) bubble in Room Temperature Ionic Liquids (RTILs) showed some phenomena that were very different from them observed in other molecular liquids. Now the reason of these interesting phenomena is being understood. The structure caused by the ionic interaction between anions and cations can exist even at the higher temperatures than the melting temperatures. It was the reason why the oscillation of Ps bubble was observed at the temperatures near the melting temperatures in RTILs. The temperature dependence of the Ps bubble size estimated by the triplet Ps (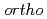 -Ps) pick-off annihilation lifetime indicated that there was a nano-meter structure remained by ionic interactions even at high temperatures. The
-Ps) pick-off annihilation lifetime indicated that there was a nano-meter structure remained by ionic interactions even at high temperatures. The 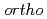 -Ps pick-off annihilation lifetime will be a strong tool to study nano-structure in RTILs.
-Ps pick-off annihilation lifetime will be a strong tool to study nano-structure in RTILs.
Hirade, Tetsuya
AIP Conference Proceedings 2182, p.030007_1 - 030007_5, 2019/12
Times Cited Count:2 Percentile:84.29(Nuclear Science & Technology)There are many new application of room temperature ionic liquids (RTILs) for many different fields. One of them is reprocessing spent nuclear fuels and then research of irradiation effects on RTILs are very important. The positron annihilation techniques are very strong tool to investigate fast reactions of excess electrons. Positron annihilation age-momentum correlation (AMOC) experiment was applied to deepen understanding the phenomena occurred at very young positron ages. We clarified that Ps bubble formation in RTILs was very slow and, finally, oscillation of Ps bubble was observed just after the Ps formation at near melting temperatures. These results can indicate that Ps formation occurred in very short time in RTILs. The phenomena observed by Ps just in RTILs will be discussed.
Hirade, Tetsuya
Yodenshi Kagaku, (11), p.33 - 40, 2018/09
The positron injected in insulating materials thermalizes at the end part of its track and forms Positronium (Ps) with one of excess electrons within  1 ps. Therefore, Ps formation can be a tool to investigate very fast processes like excess electron solvation in liquids. Many phenomena appeared in radiation chemistry researches for room temperature ionic liquids (ILs) are interesting and important for the application. Positron annihilation lifetime (PAL) measurements in ILs were carried out and anomalously long lifetime values of the shortest lifetime component were found in ILs. PAL and positron annihilation age-momentum correlation (AMOC) measurements were performed to clarify the reason of these anomalously long lifetime values and Ps bubble oscillation was finally discovered. Recent progresses are introduced with showing some results.
1 ps. Therefore, Ps formation can be a tool to investigate very fast processes like excess electron solvation in liquids. Many phenomena appeared in radiation chemistry researches for room temperature ionic liquids (ILs) are interesting and important for the application. Positron annihilation lifetime (PAL) measurements in ILs were carried out and anomalously long lifetime values of the shortest lifetime component were found in ILs. PAL and positron annihilation age-momentum correlation (AMOC) measurements were performed to clarify the reason of these anomalously long lifetime values and Ps bubble oscillation was finally discovered. Recent progresses are introduced with showing some results.
Hirade, Tetsuya
Journal of Physics; Conference Series, 618(1), p.012004_1 - 012004_5, 2015/06
Times Cited Count:5 Percentile:84.11(Physics, Applied)Positronium (Ps; a bound state of positron and electron) has a negative work function in materials and forms a bubble state in liquids. On the way of stable bubble state, the bubble grows and probably oscillates. The usual fast bubble formation has not been observed with the time resolution of the positron annihilation methods that is usually 100-200 pico-seconds. There had been many anomalous phenomena observed in the room temperature ionic liquids (RTIL). Recently it was found that very slow bubble formation caused these anomalous phenomena. And hence the oscillation of the Ps bubble was successfully observed by the change of the annihilation rate of the triplet Ps that is affected by the size of the bubble. The oscillation and the decay of the bubble give the information of the viscoelastic properties at the sub-nano scale. The temperature dependence of the oscillation has been also successfully indicated.
Matsumoto, Kazuhiko*; Hagiwara, Rika*; Ito, Yasuhiko*; Kohara, Shinji*; Suzuya, Kentaro
Nuclear Instruments and Methods in Physics Research B, 199, p.29 - 33, 2003/01
Times Cited Count:34 Percentile:88.64(Instruments & Instrumentation)The structure of 1-ethyl-3-methylimidazolium bifluoride (EMImF HF) melt has been analyzed at 333 K by a high-energy synchrotron X-ray diffraction method. The total correlation function of the EMImF
HF) melt has been analyzed at 333 K by a high-energy synchrotron X-ray diffraction method. The total correlation function of the EMImF HF melt was similar to that of the solid state, indicating that not only the short-range but also the intermediate-range ordering in the solid are partially preserved in the liquid state. The intra-molecular F-F correlation in the anions clearly appears in the total correlation function of the EMImF
HF melt was similar to that of the solid state, indicating that not only the short-range but also the intermediate-range ordering in the solid are partially preserved in the liquid state. The intra-molecular F-F correlation in the anions clearly appears in the total correlation function of the EMImF HF melt, whereas prominent peaks are not observed in the case of a room temperature molten salt, 1-ethyl-3-methylimidazolium fluorohydrogenate (EMImF
HF melt, whereas prominent peaks are not observed in the case of a room temperature molten salt, 1-ethyl-3-methylimidazolium fluorohydrogenate (EMImF 2.3HF).
2.3HF).
 single crystals with helium and Xenon ions at RT and 923K
single crystals with helium and Xenon ions at RT and 923KHojo, Kiichi; Hojo, Tomohiro; Sasajima, Naohiko*; Shirasu, Noriko; Yamashita, Toshiyuki; Minato, Kazuo; Furuno, Shigemi*
AIP Conference Proceedings 680, p.647 - 652, 2003/00
no abstracts in English
Hagiwara, Rika*; Matsumoto, Kazuhiko*; Tsuda, Tetsuya*; Ito, Yasuhiko*; Kohara, Shinji*; Suzuya, Kentaro; Matsumoto, Hajime*; Miyazaki, Yoshinori*
Journal of Non-Crystalline Solids, 312-314, p.414 - 418, 2002/10
Times Cited Count:32 Percentile:85.69(Materials Science, Ceramics)The structures of a series of XF 2.3HF (X= 1-methylimidazolium (MI), 1-ethyl-3-methylimidazolium (EMI), 1-butyl-3-methylimidazolium (BMI), 1-hexyl-3-methyl-imidazolium (HMI)) room temperature molten salts have been investigated by high-energy synchrotron X-ray diffraction technique. The correlation peaks appeared in the total correlation function are mainly ascribed to intra-molecular correlation of alkylimidazolium cations. However, it is suggested that the peak at around 3.6
2.3HF (X= 1-methylimidazolium (MI), 1-ethyl-3-methylimidazolium (EMI), 1-butyl-3-methylimidazolium (BMI), 1-hexyl-3-methyl-imidazolium (HMI)) room temperature molten salts have been investigated by high-energy synchrotron X-ray diffraction technique. The correlation peaks appeared in the total correlation function are mainly ascribed to intra-molecular correlation of alkylimidazolium cations. However, it is suggested that the peak at around 3.6  is ascribed not only to intra-molecular but also inter-molecular correlation of the cation. The contribution of the latter is also supported by the first sharp diffraction peak of the total structure factor found at almost the same position as that of a Bragg peak in the simulated X-ray diffraction pattern of solid EMIF
is ascribed not only to intra-molecular but also inter-molecular correlation of the cation. The contribution of the latter is also supported by the first sharp diffraction peak of the total structure factor found at almost the same position as that of a Bragg peak in the simulated X-ray diffraction pattern of solid EMIF HF with a layered structure, corresponding to the layer separation of
HF with a layered structure, corresponding to the layer separation of
Sato, Haruo
JNC TN8400 99-060, 12 Pages, 1999/10
Apparent diffusion coefficients(Da) of Cs(Cs ), Ni(Ni
), Ni(Ni ) and Se(SeO
) and Se(SeO
 ) in a Na-bentonite (Kunigel-V1) were measured for a dry density of 1.8 Mg
) in a Na-bentonite (Kunigel-V1) were measured for a dry density of 1.8 Mg m
m with silica sand of 30 wt% at room temperature by in-diffusion method to evaluate the effect of the mixture of silica sand on Da in bentonite. The experiments for Cs and Ni were carried out under aerobic condition, but those for Se which is redox sensitive were carried out in an Ar glove-box (O
with silica sand of 30 wt% at room temperature by in-diffusion method to evaluate the effect of the mixture of silica sand on Da in bentonite. The experiments for Cs and Ni were carried out under aerobic condition, but those for Se which is redox sensitive were carried out in an Ar glove-box (O concentration
concentration  0.1 ppm). Consequently, no significant effect of silica sand mixture to the bentonite on Da values of Cs and Se was found, and the obtained Da values were approximately the same as those in the system without silica sand reported so far. On the other hand, Da values of Ni in bentonite with silica sand were 2 orders of magnitude lower than those in bentonite without silica sand obtained to date. The Da values of Ni reported so far were obtained using stable isotopic tracer and a tracer solution with fairly high Ni concentration compared with concentration used in this study was introduced. Additionally, it is known that distribution coefficient (Kd) of Ni on Na-montmorillonite which is the major constituent clay mineral of Kunigel-V1 decreases with increasing Ni concentration. Based on this, the abrupt decrease in Da values of Ni for bentonite with silica sand is considered to be due to the difference of sorption caused by the difference of Ni concentration in the porewater of bentonite.
0.1 ppm). Consequently, no significant effect of silica sand mixture to the bentonite on Da values of Cs and Se was found, and the obtained Da values were approximately the same as those in the system without silica sand reported so far. On the other hand, Da values of Ni in bentonite with silica sand were 2 orders of magnitude lower than those in bentonite without silica sand obtained to date. The Da values of Ni reported so far were obtained using stable isotopic tracer and a tracer solution with fairly high Ni concentration compared with concentration used in this study was introduced. Additionally, it is known that distribution coefficient (Kd) of Ni on Na-montmorillonite which is the major constituent clay mineral of Kunigel-V1 decreases with increasing Ni concentration. Based on this, the abrupt decrease in Da values of Ni for bentonite with silica sand is considered to be due to the difference of sorption caused by the difference of Ni concentration in the porewater of bentonite.
Hishinuma, Akimichi; Tabuchi, Masayuki; Sawai, Tomotsugu
Intermetallics, 7(8), p.875 - 879, 1999/00
Times Cited Count:10 Percentile:59.54(Chemistry, Physical)no abstracts in English
*; Suzuya, Kentaro; Kohara, Shinji*; *; L.A.Curtiss*; Saboungi, M. L.*
Zeitschrift f r Physikalische Chemie, 209, p.209 - 221, 1999/00
r Physikalische Chemie, 209, p.209 - 221, 1999/00
no abstracts in English
S.M.Spiessberger*; K.Humer*; H.W.Weber*; E.K.Tschegg*; H.Gerstenberg*; Udagawa, Akira
Advances in Cryogenic Engineering Materials, Vol.44, p.191 - 195, 1998/00
no abstracts in English
Hojo, Kiichi; Furuno, Shigemi; Kushita, Kohei; *; *; *
Nuclear Instruments and Methods in Physics Research B, 116, p.382 - 388, 1996/00
Times Cited Count:32 Percentile:91(Instruments & Instrumentation)no abstracts in English
Hojo, Kiichi; Furuno, Shigemi; *; Kushita, Kohei; ; *
Microsc. Microanal. Microstruct., 6, p.141 - 147, 1995/02
Times Cited Count:2 Percentile:23.42(Microscopy)no abstracts in English
 and H
and H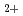 dual-ion beam irradiation
dual-ion beam irradiationHojo, Kiichi; Furuno, Shigemi; Kushita, Kohei; ; *
Journal of Nuclear Materials, 191-194, p.583 - 587, 1992/00
Times Cited Count:25 Percentile:88.48(Materials Science, Multidisciplinary)no abstracts in English
; ; Kondo, Tatsuo
JAERI-M 82-052, 31 Pages, 1982/06
no abstracts in English
Nuclear Instruments and Methods, 196(1), p.121 - 130, 1982/00
no abstracts in English
Proc.1981 INS Int.Symp.on Nuclear Radiation Detectors, p.159 - 177, 1981/00
no abstracts in English